Electrolysis Of Molten
Compounds
Process of breaking down compounds in molten
or aqueous state into their constituent elements by
passing electricity through them.
v
Anode
(Positive electrode)
─
Negative ions or anions
are attracted to anode.
─
Anion undergo discharge by releasing electrons to form neutral
atoms.
v Cathode
(Negative electrode)
─
Positive ions or cations
are attracted to cathode.
─
Cation undergo discharge to form atoms.
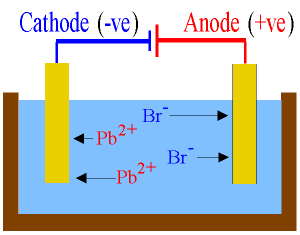
Electrolysis Of Molten Lead (II) Bromide
v
Anode (Positive electrode)
─
Anions,
Br - are attracted
to the anode.
─
Bromide
ions, Br- discharge by releasing
electron to form bromine gas,
Br2
Half equation :
2Br-(l)
→ Br2(g) + 2e-
v
Cathode (negative electrodes)
─
Lead(II)
ions, Pb2+ are attracted
to the cathode.
─
Lead
(II) ions, Pb2+ undergo discharge
by accepting two electrons to
form a lead atom/lead metal.
Half equation :
Pb2+ (I) + 2e- → Pb (s)
Overall equation:
Pb2+ (I) + 2Br - → Pb(s) + Br2(g)
I have problem with the question in past year ques book pg 231 ques num 5(e). Im confused with its electrons flow and terminals.
ReplyDelete